
FDA gives nod to first fully-removable percutaneous peripheral
Price: $ 79.99
4.9(733)
SPR Therapeutics has developed a peripheral nerve stimulation system that is placed percutaneously through the skin instead of being implanted and which can be completely removed from the body after therapy period.
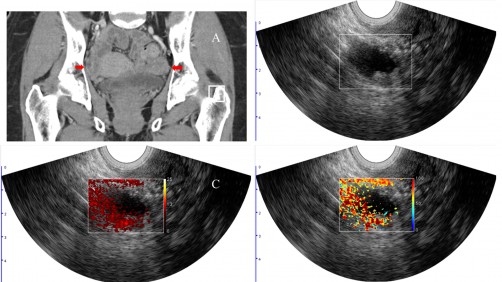
News - Interventional News
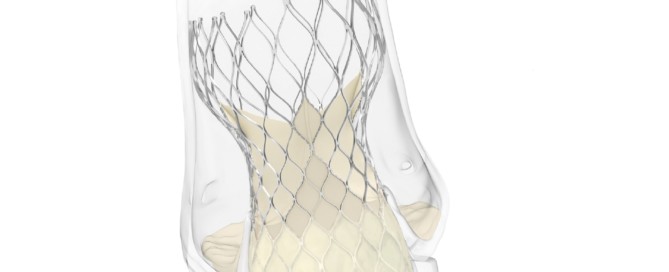
Approvals & refusals – BIBA Medtech Insights

Latest News Archives - Page 2 of 3 - Interventional News

willdate, Author at Cardiac Rhythm News

FDA approves Nevro's Senza system to treat chronic pain with diabetic neuropathy - MassDevice
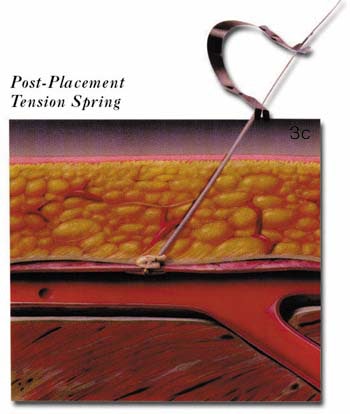
Arterial puncture site management

Microparticles: biogenesis, characteristics and intervention therapy for cancers in preclinical and clinical research, Journal of Nanobiotechnology

NeuroNews issue 50 - US by BIBA Publishing - Issuu

Design principles of microneedles for drug delivery and sampling applications - ScienceDirect

NeuroNews issue 50 - US by BIBA Publishing - Issuu

Role of Immunological Cells in Hepatocellular Carcinoma Disease and Associated Pathways

CFR 2022 – Volume 8 by Radcliffe Cardiology - Issuu

Percutaneous peripheral nerve stimulation for the treatment of chronic neuropathic postamputation pain: a multicenter, randomized, placebo-controlled trial




